PUBLICATION NEWS

22.11.2018
Our fellow Alessio Terenzi has published as co-author in Scientific Reports: “Biological activity of PtIV prodrugs triggered by riboflavin-mediated bioorthogonal photocatalysis”.
The new study is a joint effort between the group of Prof. Luca Salassa (Donostia International Physics Center) and the groups of Profs. Walter Berger and Bernhard K. Keppler (Research Platform “Translational Cancer Therapy Research”, University of Vienna and Medical University of Vienna). The study highlights the possibility of combining photocatalysis and Pt(IV) prodrugs in the treatment of cancer. Congratulations!!
Abstract:
We have recently demonstrated that riboflavin (Rf) functions as unconventional bioorthogonal photocatalyst for the activation of PtIV prodrugs. In this study, we show how the combination of light and Rf with two PtIV prodrugs is a feasible strategy for light-mediated pancreatic cancer cell death induction. In Capan-1 cells, which have high tolerance against photodynamic therapy, Rf-mediated activation of the cisplatin and carboplatin prodrugs cis,cis,trans-[Pt(NH3)2(Cl)2(O2CCH2CH2CO2H)2] (1) and cis,cis,trans-[Pt(NH3)2(CBDCA)(O2CCH2CH2CO2H)2] (2, where CBDCA = cyclobutane dicarboxylate) resulted in pronounced reduction of the cell viability, including under hypoxia conditions. Such photoactivation mode occurs to a considerable extent intracellularly, as demonstrated for 1 by uptake and cell viability experiments. 195Pt NMR, DNA binding studies using circular dichroism, mass spectrometry and immunofluorescence microscopy were performed using the Rf-1 catalyst-substrate pair and indicated that cell death is associated with the efficient light-induced formation of cisplatin. Accordingly, Western blot analysis revealed signs of DNA damage and activation of cell death pathways through Rf-mediated photochemical activation. Phosphorylation of H2AX as indicator for DNA damage, was detected for Rf-1 in a strictly light-dependent fashion while in case of free cisplatin also in the dark. Photochemical induction of nuclear pH2AX foci by Rf-1 was confirmed in fluorescence microscopy again proving efficient light-induced cisplatin release from the prodrug system.
NEW PUBLICATION

NEW PUBLICATION
15.11.2018
Our fellow Nicola Silva has published as co-author in PLOS Genetics: “C. elegans ZHP-4 is required at multiple distinct steps in the formation of crossovers and their transition to segregation competent chiasmata”. Congratulations Nicola!
Abstract:
Correct segregation of meiotic chromosomes depends on DNA crossovers (COs) between homologs that culminate into visible physical linkages called chiasmata. COs emerge from a larger population of joint molecules (JM), the remainder of which are repaired as noncrossovers (NCOs) to restore genomic integrity. We present evidence that the RNF212-like C. elegans protein ZHP-4 cooperates with its paralog ZHP-3 to enforce crossover formation at distinct steps during meiotic prophase: in the formation of early JMs and in transition of late CO intermediates into chiasmata. ZHP-3/4 localize to the synaptonemal complex (SC) co-dependently followed by their restriction to sites of designated COs. RING domain mutants revealed a critical function for ZHP-4 in localization of both proteins to the SC and for CO formation. While recombination initiates in zhp-4 mutants, they fail to appropriately acquire pro-crossover factors at abundant early JMs, indicating a function for ZHP-4 in an early step of the CO/NCO decision. At late pachytene stages, hypomorphic mutants exhibit significant levels of crossing over that are accompanied by defects in localization of pro-crossover RMH-1, MSH-5 and COSA-1 to designated crossover sites, and by the appearance of bivalents defective in chromosome remodelling required for segregation. These results reveal a ZHP-4 function at designated CO sites where it is required to stabilize pro-crossover factors at the late crossover intermediate, which in turn are required for the transition to a chiasma that is required for bivalent remodelling. Our study reveals an essential requirement for ZHP-4 in negotiating both the formation of COs and their ability to transition to structures capable of directing accurate chromosome segregation. We propose that ZHP-4 acts in concert with ZHP-3 to propel interhomolog JMs along the crossover pathway by stabilizing pro-CO factors that associate with early and late intermediates, thereby protecting designated crossovers as they transition into the chiasmata required for accurate disjunction.
NEW PUBLICATION
NEW PUBLICATION
06.11.2018
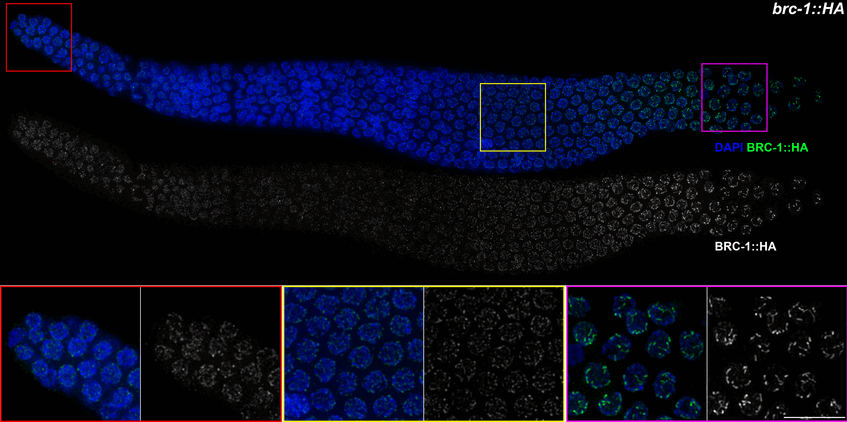
The INDICAR-Fellow Nicola Silva has published as corresponding author in PLOS Genetics: “BRCA1-BARD1 associate with the synaptonemal complex and pro-crossover factors and influence RAD-51 dynamics during Caenorhabditis elegans meiosis”. Congratulations Nicola!!
Abstract:
During meiosis, the maternal and paternal homologous chromosomes must align along their entire length and recombine to achieve faithful segregation in the gametes. Meiotic recombination is accomplished through the formation of DNA double-strand breaks, a subset of which can mature into crossovers to link the parental homologous chromosomes and promote their segregation. Breast and ovarian cancer susceptibility protein BRCA1 and its heterodimeric partner BARD1 play a pivotal role in DNA repair in mitotic cells; however, their functions in gametogenesis are less well understood. Here we show that localization of BRC-1 and BRD-1 (Caenorhabditis elegans orthologues of BRCA1 and BARD1) is dynamic during meiotic prophase I; they ultimately becoming concentrated at regions surrounding the presumptive crossover sites, co-localizing with the pro-crossover factors COSA-1, MSH-5 and ZHP-3. The synaptonemal complex and PLK-2 activity are essential for recruitment of BRC-1 to chromosomes and its subsequent redistribution towards the short arm of the bivalent. BRC-1 and BRD-1 form in vivo complexes with the synaptonemal complex component SYP-3 and the crossover-promoting factor MSH-5. Furthermore, BRC-1 is essential for efficient stage-specific recruitment/stabilization of the RAD-51 recombinase to DNA damage sites when synapsis is impaired and upon induction of exogenous damage. Taken together, our data provide new insights into the localization and meiotic function of the BRC-1–BRD-1 complex and highlight its essential role in DNA double-strand break repair during gametogenesis.
INDICAR-Workshop 2018
21.09.2017
The INDICAR-Workshop 2018 took place in Bratislava (Slovakia), from 13th to 14th Sep. The INDICAR Workshop is the opportunity for post-doctoral researchers enrolled in the INDICAR Program to discuss common themes and exchange cross-disciplinary results with a particular focus on the techniques used to reach their goals. As the INDICAR Programme ends November 2018, the fellows presented and discussed their work with a particular focus on how to wrap up outcomes and what the results may mean for future research.


ANTJE KOLLER’S VIDEO ON BREAKTHROUGH CANCER PAIN

ANTJE KOLLER’S VIDEO ON BREAKTHROUGH CANCER PAIN
21.08.2018
Our fellow Antje Koller took part in an educational video discussing the latest advances on breakthrough cancer pain (BTcP). The video is aired on ecancer tv, an organization devoted to provide educational content free to the global oncology community.
Dr Koller discusses together with the host, Dr Andrew Davies, and the rest of the panel members, Dr Didier Mayeur and Dr Jason Boland, the latest perspectives on BTcP, its impact on patients’ lives, the current strategies and the importance of individualized treatments and multi-disciplinary approaches.
Please notice that watching the video requires previous registration on ecancer.org.
NEW PUBLICATION
NEW PUBLICATION
09.08.2018
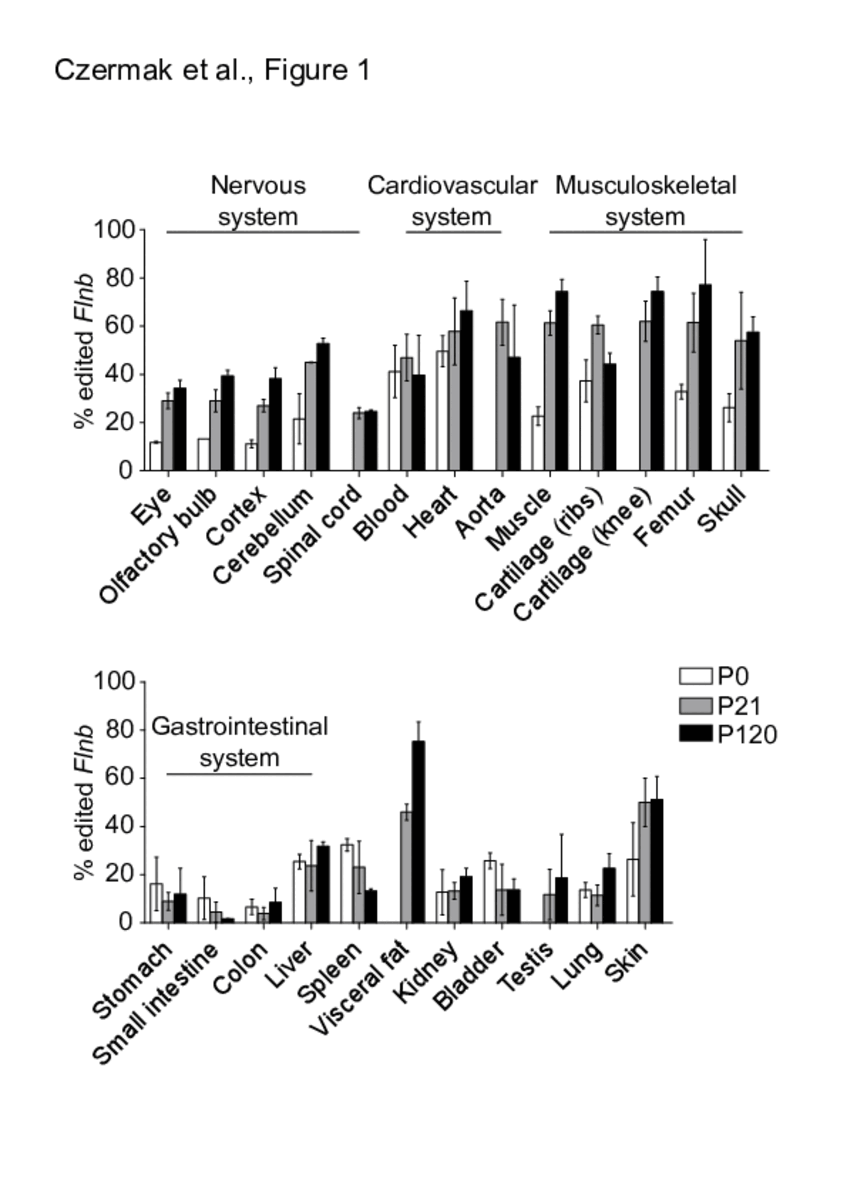
The INDICAR-Fellow Laura Cimatti has published as last author in the Journal RNA Biology: “Organ-wide profiling in mouse reveals high editing levels of Filamin B mRNA in the musculoskeletal system”. Congratulations Laura!
Abstract:
Adenosine to inosine RNA editing in protein-coding messenger RNAs (mRNAs) potentially leads to changes in the amino acid composition of the encoded proteins. The mRNAs encoding the ubiquitously expressed actin-crosslinking proteins Filamin A and Filamin B undergo RNA editing leading to a highly conserved glutamine to arginine exchange at the identical position in either protein. Here, by targeted amplicon sequencing we analysed the RNA editing of Filamin B across several mouse tissues during post-natal development. We find highest filamin B editing levels in skeletal muscles, cartilage and bones, tissues where Filamin B function seems most important. Through the analysis of Filamin B editing in mice deficient in either ADAR1 or 2, we identified ADAR2 as the enzyme responsible for Filamin B RNA editing. We show that in neuronal tissues Filamin B editing drops in spliced transcripts indicating regulated maturation of edited transcripts. We show further that the variability of Filamin B editing across several organs correlates with its mRNA expression.
NEW PUBLICATION
03.08.2018
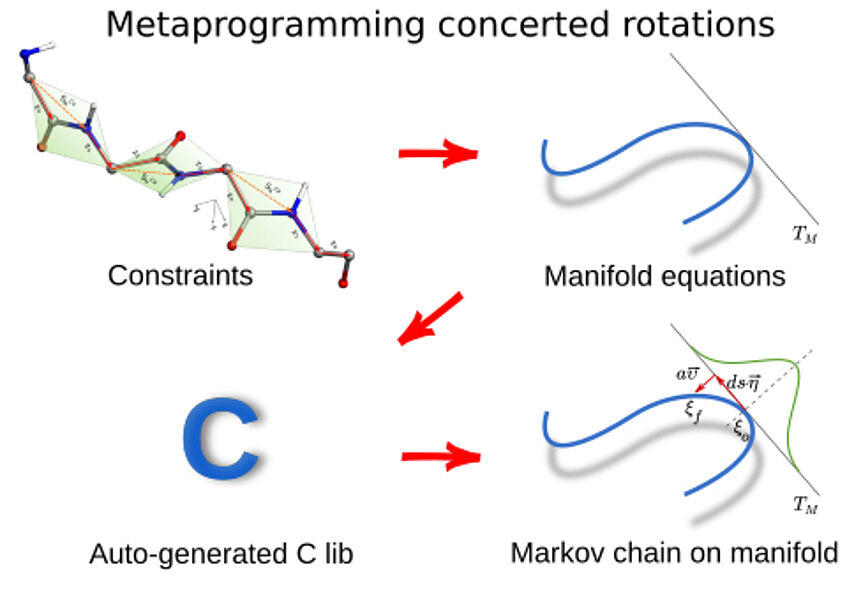
Our fellow Luca Tubiana just published as corresponding author in the European Physical Journal E Soft Matter: “Implementing efficient concerted rotations using Mathematica and C code⋆”. Congratulations Luca!
Abstract:
In this article we demonstrate a general and efficient metaprogramming implementation of concerted rotations using Mathematica. Concerted rotations allow the movement of a fixed portion of a polymer backbone with fixed bending angles, like a protein, while maintaining the correct geometry of the backbone and the initial and final points of the portion fixed. Our implementation uses Mathematica to generate a C code which is then wrapped in a library by a Python script. The user can modify the Mathematica notebook to generate a set of concerted rotations suited for a particular backbone geometry, without having to write the C code himself. The resulting code is highly optimized, performing on the order of thousands of operations per second.
NEW PUBLICATION

NEW PUBLICATION
30.07.2018
The INDICAR-Fellow Luca Tubiana has published as corresponding author in the European Physical Journal E Soft Matter: “KymoKnot: A web server and software package to identify and locate knots in trajectories of linear or circular polymers”, and his research article has been featured on the cover! Congratulations Luca!!
Abstract:
The KymoKnot software package and web server identifies and locates physical knots or proper knots in a series of polymer conformations. It is mainly intended as an analysis tool for trajectories of linear or circular polymers, but it can be used on single instances too, e.g. protein structures in PDB format. A key element of the software package is the so-called minimally interfering chain closure algorithm that is used to detect physical knots in open chains and to locate the knotted region in both open and closed chains. The web server offers a user-friendly graphical interface that identifies the knot type and highlights the knotted region on each frame of the trajectory, which the user can visualize interactively from various viewpoints. The dynamical evolution of the knotted region along the chain contour is presented as a kymograph. All data can be downloaded in text format. The KymoKnot package is licensed under the BSD 3-Clause licence. The server is publicly available at http://kymoknot.sissa.it/kymoknot/interactive.php.
NEW PUBLICATION
25.07.2018
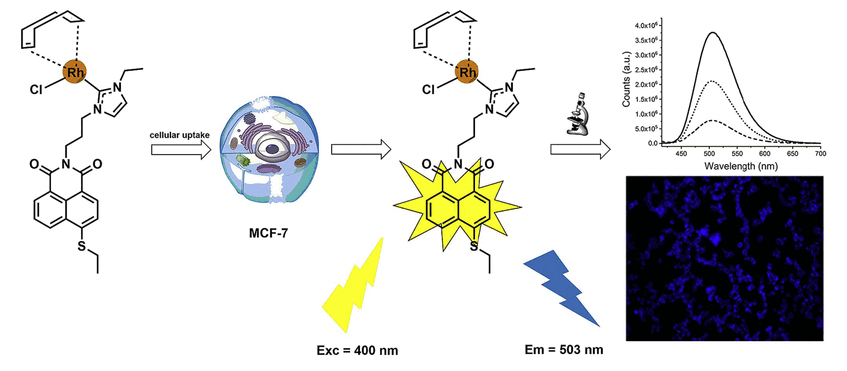
Our fellow Alessio Terenzi has published as co-author in the European Journal of Medicinal Chemistry: “Fluorescent organometallic rhodium(I) and ruthenium(II) metallodrugs with 4-ethylthio-1,8-naphthalimide ligands: Antiproliferative effects, cellular uptake and DNA-interaction”. Congratulations!
Abstract:
Fluorescent 4-ethylthio-1,8-naphthalimides containing rhodium(I) N-heterocyclic carbene (NHC) and ruthenium (II) NHC fragments were synthesised and evaluated for their antiproliferative effects, cellular uptake and DNA-binding activity. Both types of organometallics triggered ligand dependent efficient cytotoxic effects against tumor cells with the rhodium(I) NHC derivatives causing stronger effects than the ruthenium (II) NHC analogues. Antiproliferative effects could also be observed against several pathogenic Gram-positive bacterial strains, whereas the growth of Gram-negative bacteria was not substantially affected. Cellular uptake was confirmed by atomic absorption spectroscopy as well as by fluorescence microscopy indicating a general ligand dependent accumulation in the cells. An in-depth study on the interaction with DNA confirmed insertion of the naphthalimide moiety between the planar bases of B-DNA via an intercalation mechanism, as well as its stacking on top of the quartets of G-quadruplex structures. Furthermore, additional coordinative binding of the organometallic complexes to the model DNA base 9-ethylguanine could be detected. The studied compounds thus represent promising bioorganometallics featuring strong pharmacological effects in combination with excellent cellular imaging properties.
NEW PUBLICATION
16.07.2018
The INDICAR-Fellow Alessio Terenzi co-authored a publication in the European Journal of Inorganic Chemistry: “Synthesis and Biological Evaluation of Organometallic Complexes Bearing Bis‐1,8‐naphthalimide Ligands”. Congratulations Alessio!
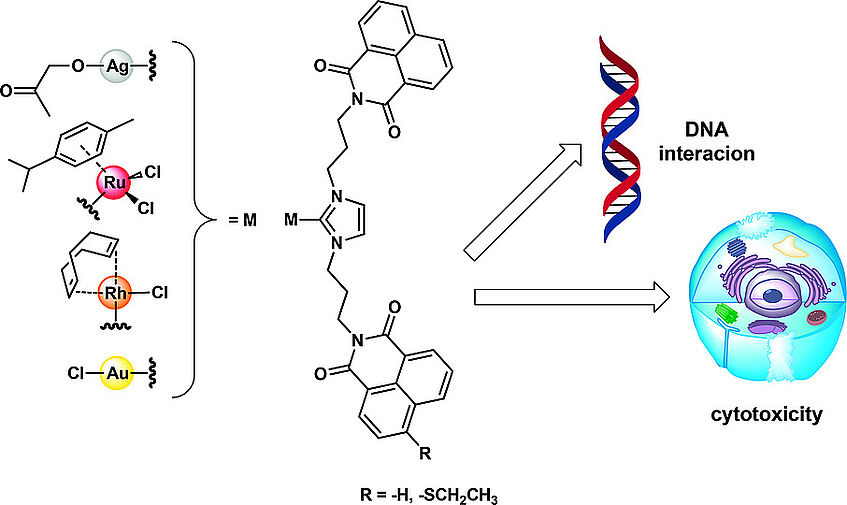
Abstract:
Organometallic N‐heterocyclic carbene (NHC) complexes with intercalating bis‐naphthalimide ligands were prepared and evaluated biologically. Cytotoxic effects against tumor cells or bacteria were strongly ligand dependent with minor influence of the metal (Ag, Ru, Rh, Au) centers. Complex 8b with a rhodium(I) NHC moiety was studied in more detail for its DNA interacting properties in comparison to the metal free ligand. These studies showed a good DNA binding pattern with some preference for the telomeric quadruplex structure hTelo. Complex 8b was also shown to trigger additional coordinative binding to the DNA and therefore represents an useful tool compound with a mixed intercalative/coordinative DNA binding mode.
NEW PUBLICATION

NEW PUBLICATION
22.06.2018
The INDICAR-Fellow Nicola Silva co-authored a PLoS Genetics publication: “The conserved LEM-3/Ankle1 nuclease is involved in the combinatorial regulation of meiotic recombination repair and chromosome segregation in Caenorhabditis elegans”. Congratulations Nicola!
Abstract:
Homologous recombination is essential for crossover (CO) formation and accurate chromosome segregation during meiosis. It is of considerable importance to work out how recombination intermediates are processed, leading to CO and non-crossover (NCO) outcome. Genetic analysis in budding yeast and Caenorhabditis elegans indicates that the processing of meiotic recombination intermediates involves a combination of nucleases and DNA repair enzymes. We previously reported that in C. elegans meiotic joint molecule resolution is mediated by two redundant pathways, conferred by the SLX-1 and MUS-81 nucleases, and by the HIM-6 Bloom helicase in conjunction with the XPF-1 endonuclease, respectively. Both pathways require the scaffold protein SLX-4. However, in the absence of all these enzymes, residual processing of meiotic recombination intermediates still occurs and CO formation is reduced but not abolished. Here we show that the LEM-3 nuclease, mutation of which by itself does not have an overt meiotic phenotype, genetically interacts with slx-1 and mus-81 mutants, the respective double mutants displaying 100% embryonic lethality. The combined loss of LEM-3 and MUS-81 leads to altered processing of recombination intermediates, a delayed disassembly of foci associated with CO designated sites, and the formation of univalents linked by SPO-11 dependent chromatin bridges (dissociated bivalents). However, LEM-3 foci do not colocalize with ZHP-3, a marker that congresses into CO designated sites. In addition, neither CO frequency nor distribution is altered in lem-3 single mutants or in combination with mus-81 or slx-4 mutations. Finally, we found persistent chromatin bridges during meiotic divisions in lem-3; slx-4 double mutants. Supported by the localization of LEM-3 between dividing meiotic nuclei, this data suggest that LEM-3 is able to process erroneous recombination intermediates that persist into the second meiotic division.
KONSTANTINOS KIAKOS PRESENTED HIS VIDEO “DRUGGING CANCER” AT TOP KINO
19.06.2018
On the 7th of June the INDICAR Fellow Konstantinos Kiakos presented his video “Drugging Cancer” at Top Kino, together with participants of the CommunicationHub.
Last October Konstantinos enrolled the CommunicationHub 2017/18 program "My research as video", organized by Knowledge Transfer Center East (WTZ Ost). Konstantinos and his team worked on his video under the guidance and support of experts from Okto TV.
“Drugging Cancer” aims at disseminating the INDICAR program and Konstantinos’ research and to raise awareness on the research performed to discover new cancer drugs.
THE EUTOPIA PROJECT LEAD BY LUCA TUBIANA HAS BEEN AWARDED A COST GRANT
29.05.2018
The EUTOPIA (EUropean TOPology Interdisciplinary Action) project lead by INDICAR Fellow Luca Tubiana has been awarded a COST grant. COST is an EU-funded program that provides funding and support to researchers to establish interdisciplinary networks in Europe and beyond. EUTOPIA aims at forming an EU community of researchers from different fields working on the topological aspects of soft and bio-materials. The network of EUTOPIA members span 11 European countries. Thanks to the COST grant, EUTOPIA members will be able to perform scientific exchanges and organize meetings, conferences, training schools or other scientific networking activities.

TALKS AT VIENNA EUROPEAN SCHOOL
11.04.2018
On Wednesday 21st of March, the INDICAR Fellows Laura Cimatti and Mohamed Elgendy visited the Vienna European School. They presented grade 5 and 6 students interactive and engaging talks about the everyday life of a scientist and their research. This initiative aims to create awareness among secondary students of the research performed by INDICAR Fellows and to develop their interest in research careers. Thanks Vienna European School for having us, it was a great experience!



NEW PUBLICATION
28.03.2018
Our fellow Takahiko Akematsu published as corresponding author in the journal Genes: “Resistance to 6-Methylpurine is Conferred by Defective Adenine Phosphoribosyltransferase in Tetrahymena”. Congratulations Takahiko!
6-methylpurine (6mp) is a toxic analog of adenine that inhibits RNA and protein synthesis and interferes with adenine salvage mediated by adenine phosphoribosyltransferase (APRTase). Mutants of the ciliated protist Tetrahymena thermophila that are resistant to 6mp were isolated in 1974, but the mechanism of resistance has remained unknown. To investigate 6mp resistance in T. thermophila, we created 6mp-resistant strains and identified a mutation in the APRTase genomic locus (APRT1) that is responsible for 6mp resistance. While overexpression of the mutated APRT1 allele in 6mp-sensitive cells did not confer resistance to 6mp, reduced wild-type APRT1 expression resulted in a significant decrease in sensitivity to 6mp. Knocking out or reducing the expression of APRT1 by RNA interference (RNAi) did not affect robust cell growth, which indicates that adenine salvage is redundant or that de novo synthesis pathways provide sufficient adenosine monophosphate for viability. We also explored whether 6mp resistance could be used as a novel inducible selection marker by generating 6mp- and paromomycin-resistant double mutants. While 6mp- and paromomycin-resistant double mutants did express fluorescent proteins in an RNAi-based system, the system requires optimization before 6mp resistance can be used as an effective inducible selection marker.
KONSTANTINOS KIAKOS’ RESEARCH ON VIDEO
26.03.2018
Last week, the INDICAR Fellow Konstantinos Kiakos and his team filmed a video about the INDICAR Programme, his research and its impact on society as well. Dr. Kiakos enrolled last October the CommunicationHub 2017/18 “My research as video“, organized by the Knowledge Transfer Center East (WTZ Ost). As part of this programme, he has received support and guidance from experts from Okto TV to create his video.
Dr. Kiakos recently filed a U.S. provisional patent on the therapeutic composition and methods of use based on a series of novel STAT3 small-molecule Inhibitors.

ANNE CONIBEAR PUBLICATION FEATURED ON THE COVER OF CHEMBIOCHEM
26.02.2018
Anne Conibear research article “Synthetic cancer-targeting innate immune stimulators give insights into avidity effects”, published in the journal ChemBioChem, has been featured on the cover. Congratulations Anne!

GOOD BYE FIRST-CALL FELLOWS, WE WILL MISS YOU!
23.02.18
Last Monday INDICAR bade farewell to its fellows from the first cohort. The event gathered supervisors, fellows, peers, and colleagues to thank their support to the INDICAR Programme and to celebrate INDICAR Fellows’ accomplishments.
We wish our fellows all the best in their careers!

CONGRATULATIONS TO ANTJE KOLLER ON BEING AWARDED THE "BACK-TO-RESEARCH" GRANT
29.01.2018
We are delighted to announce that our fellow Antje Koller have been awarded the "Back-to-Research" Grant, from the University of Vienna.
The funding scheme is addressed to female postdoctoral researchers who, during the last five years, have reduced or interrupted their research because of care obligations in their families. The grant provides researchers with the possibility to complete research applications and/or publications in order to ensure that they can re-enter or continue their academic careers.

NEW PUBLICATION
22.01.2018
The INDICAR-Fellow Anne Conibear published the paper “Recent Advances in Peptide-Based Approaches for Cancer Treatment”, in the journal Current Medicinal Chemistry. Congratulations Anne!
Peptide-based pharmaceuticals have recently experienced a renaissance due to their ability to fill the gap between the two main classes of available drugs, small molecules and biologics. Peptides combine the high potency and selectivity typical of large proteins with some of the characteristic advantages of small molecules such as synthetic accessibility, stability and the potential of oral bioavailability. One of the applications in which peptide-based approaches have grown rapidly is cancer therapy, with a focus on new and established targets. Many novel peptide-based methods for cancer treatment have been developed in the last few years, ranging from naturally-occurring and modified peptides to peptide-drug conjugates, peptide nanomaterials and peptide-based vaccines. This review focuses on recent advances, advantages and challenges of these selected peptide-based approaches for cancer treatment.

KONSTANTINOS KIAKOS FILES A U.S. PROVISIONAL PATENT
10.01.2018
The INDICAR fellow Konstantinos Kiakos has filed a U.S. provisional patent on the therapeutic composition and methods of use based on a series of novel STAT3 small-molecule inhibitors. This new class of compounds provides a method for selective and effective reduction of pSTAT3 in cancer cells, leading to apoptosis. The STAT3 inhibitors described in the invention can chemo- and radio- sensitize cancer cells and reverse platinum drug resistance.

AUSTRIAN CAFÉ
30.11.2017
Yesterday, the Austrian Café took place at the Irish pub Flanagans. INDICAR fellows and friends discussed the paper presented by Alessio: “Local epigenetic reprogramming induced by G-quadruplex ligands”. With this activity, INDICAR aims to foster informal and relaxed scientific discussions. As a result, INDICAR fellows have the opportunity to network and obtain feedback about their research from peers.


NEW PUBLICATION
08.11.2017
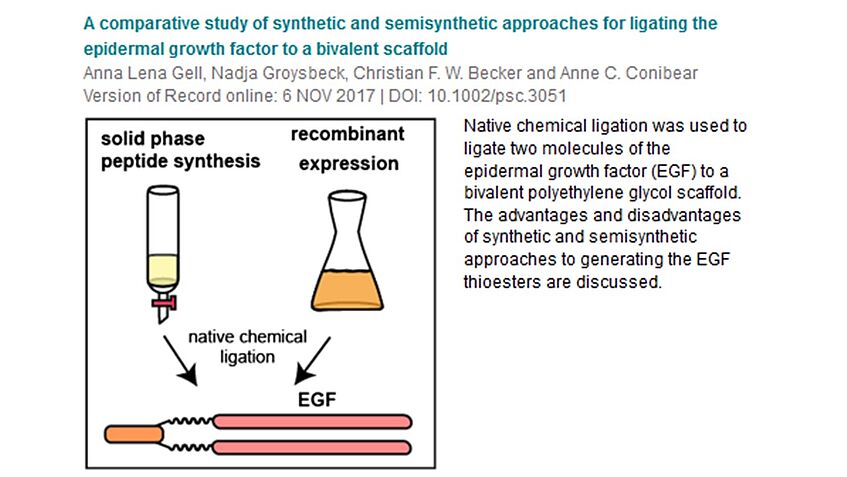
Our fellow Anne Conibear published as corresponding author in the Journal of Peptide Science: “A comparative study of synthetic and semisynthetic approaches for ligating the epidermal growth factor to a bivalent scaffold”. Congratulations Anne!
A prominent target of monoclonal antibodies as targeted therapies for cancer is the epidermal growth factor receptor, which is overexpressed on the surface of various cancer cell types. Its natural binder, the epidermal growth factor (EGF), is a 53 amino acid polypeptide. Anticancer synthetic targeted immune system engagers (ISErs) comprising two ‘binder’ peptides, which are attached to a scaffold conveying immune stimulating ‘effector’ properties, via monodisperse polyethylene glycol chains. So far, preparation of ISErs has been limited to the use of small peptides (8–20 amino acids) as binding functionalities, and they have been entirely synthesized by solid phase peptide synthesis. Here, we describe a synthetic and a semisynthetic approach for the preparation of an ISEr bearing two murine EGF molecules as binding entities (ISEr-EGF2). EGF was either synthesized in segments by solid phase peptide synthesis or expressed recombinantly and ligated to the scaffold by native chemical ligation. We report the successful generation of synthetic and semisynthetic ISEr-EGF2 as well as several challenges encountered during the synthesis and ligations. We demonstrate the application of native chemical ligation for the design of larger ISEr constructs, facilitating new objectives for the coupling of small binder peptides and larger proteins to multivalent ISEr scaffolds.
TALKS AT VIENNA INTERNATIONAL SCHOOL
30.10.2017
Last Wednesday the INDICAR-Fellow Laura Cimatti visited Vienna International School. She presented interactive and engaging talks to grade 10 and 11 students. Laura talked about her research and everyday life of scientist, and answered students’ questions. Thanks Vienna International School for having us, it was a great experience and we hope to see you again soon!

INDICAR, HEADER OF CORDIS
09.10.2017
Our fellows’ picture is the Twitter header of @CORDIS_EU, the Community Research and Development Information Service of the European Commission. Thanks!!!

NEW PUBLICATION

NEW PUBLICATION
05.10.2017
The INDICAR-Fellow Antje Koller published in the Journal of Pain and Symptom Management: “Developing a short form of the German Barriers Questionnaire-II: A validation study in four steps”. Congratulations Antje!
CONTEXT:
Patient-related barriers to cancer pain management are most commonly assessed with the Barriers Questionnaire-II (BQII; 27 items).
OBJECTIVES:
The aim of this study was to develop a valid short form of the BQII-German (BQII-G) to increase usability in clinical routines and reduce patient burden.
METHODS:
The validation study comprised a stepwise approach. In the first step, the linguistic validated version of the BQII-G was psychometrically tested for internal consistency and factor structure (N=207). The second step included an independent peer review in terms of expert ratings (4 nurses and 2 patients) of each of the BQII-G items regarding (rather) include or (rather) not include, according to the content validity index. The third step comprised a consensus process to integrate the expert ratings into a short form of the BQII-G (BQII-G12). The fourth step included a preliminary psychometric exploration of the short-version BQII-G12.
RESULTS:
Cronbach's α was .92 for the BQII-G. Steps 1 to 3 resulted in the BQII-G12 (12 items). The correlation showed that the BQII-G12 explains 84.3% (r=.92) of the variance of the BQII-G. Crohnbach's alpha of the BQII-G12 was .833.
CONCLUSIONS:
The BQII-G12 showed excellent psychometric properties in the preliminary testing, providing a new option for practice and Research.
INDICAR-Fellows attended a writing skills workshop
03.10.2017
INDICAR-Fellows attended yesterday the workshop „How to get your paper published”. The trainer, Helen Pickersgill (Life Science Editors), provided them with insights on how to write more competitive papers and get published in the top-Level Journal. During the morning session, INDICAR-Fellows learnt how to highlight the strengths and minimize the weaknesses of a paper, and how to engage the reader. The afternoon session was focused on the publication process and what the journals and editors are looking for.

INDICAR-Workshop 2017

INDICAR-Workshop 2017
20.09.2017
The INDICAR-Workshop 2017 took place in Kalamata (Greece), from Monday 11th to Thursday 14th Sep 2017. The INDICAR Workshop is the opportunity for post-doctoral researchers enrolled in the INDICAR Program to discuss common themes and exchange cross-disciplinary results with a particular focus on the techniques used to reach their goals. The event featured talks from invited guest speakers, post-doctoral researchers and PhD students. Besides, the event included in this edition talks from experts on science communication, technology transfer and industry. The workshop promoted broad discussion and interaction among participants. As a result, participants came up with new ideas and potential collaborations and start-ups in the field of cancer research.
ANNE CONIBEAR PRESENTED HER RESEARCH AT THE 7TH CHEMICAL PROTEIN SYNTHESIS MEETING
19.09.2017
The INDICAR-Fellow Anne Conibear attended the 7th Chemical Protein Synthesis Meeting that took place on Sept 4-7 in Haifa, Israel. There, she presented her poster 'Synthetic Targeted Innate Immune Stimulators to Explore Avidity Effects', Anne Conibear, Andre Pötgens and Christian Becker. Well done Anne!
NICOLA SILVA PRESENTED HIS RESEARCH AT THE EMBO CONFERENCE ON MEIOSIS
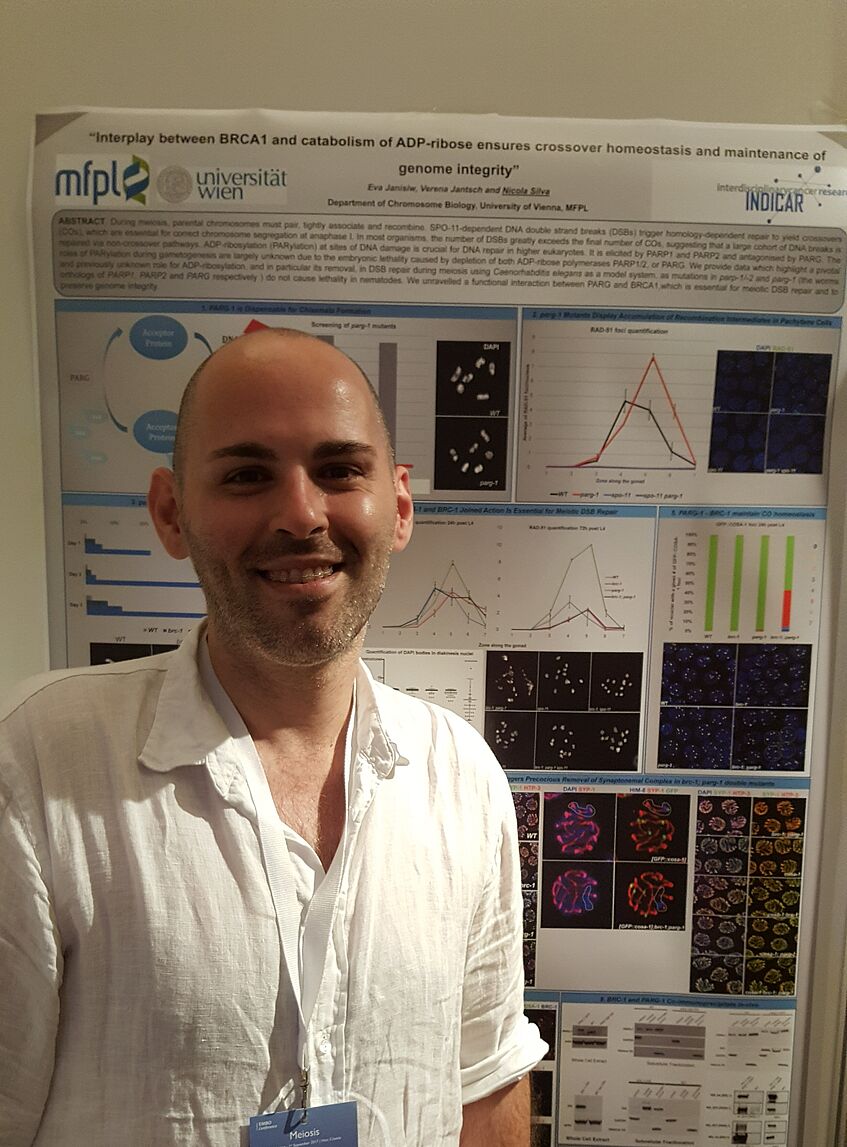
NICOLA SILVA PRESENTED HIS RESEARCH AT THE EMBO CONFERENCE ON MEIOSIS
29.08.2017
The INDICAR-Fellow Nicola Silva presented yesterday his poster: “Interplay between BRCA1 and catabolism of ADP-ribose ensures crossover homeostasis and maintenance of genome integrity” at the EMBO Conference on Meiosis in Hvar, Croatia. Well done!
NEW PUBLICATION
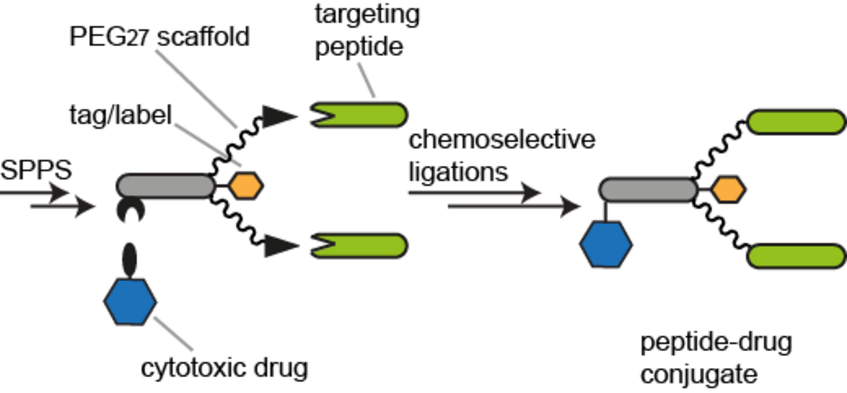
NEW PUBLICATION
21.08.2017
The INDICAR-Fellow Anne Conibear published in the journal Bioconjugate Chemistry: “Multifunctional αvβ6 integrin-specific peptide-Pt(IV) conjugates for cancer cell targeting”. Congratulations Anne!
Increasing the specificity of cancer therapy, and thereby decreasing damage to normal cells, requires targeting to cancer-cell specific features. The αvβ6 integrin is a receptor involved in cell adhesion and is frequently up-regulated in cancer cells compared to normal cells. We have selected a peptide ligand reported to bind specifically to the β6 integrin and have synthesized a suite of multispecific molecules to explore the potential for targeting of cancer cells. A combination of solid-phase peptide synthesis and chemoselective ligations was used to synthesize multifunctional molecules composed of integrin-targeting peptides, cytotoxic platinum(IV) prodrugs, and fluorescent or affinity probes joined with flexible linkers. The modular synthesis approach facilitates the construction of peptide–drug conjugates with various valencies and properties in a convergent manner. The binding and specificity of the multifunctional peptide conjugates were investigated using a cell line transfected with the β6 integrin and fluorescence microscopy. This versatile and highly controlled approach to synthesizing labeled peptide–drug conjugates has the potential to target potent cytotoxic drugs specifically to cancer cells, reducing the doses required for effective treatment.
TWO INDICAR-FELLOWS GAVE TALKS AT THE METALLOMICS CONFERENCE IN VIENNA
17.08.2017
Yesterday, two of our fellows gave talks at the Metallomics conference in Vienna. Kostas presented his research: “Restoring cellular sensitivity to platinum‐based drugs by targeted inhibition of STAT3” and Alessio: “Ruthenium arene complexes for G‐quadruplex DNA recognition”. Great job!!

LUCA TUBIANA PRESENTED HIS RESEARCH AT THE WORKSHOP ON PHYSICAL VIROLOGY IN TRIESTE

LUCA TUBIANA PRESENTED HIS RESEARCH AT THE WORKSHOP ON PHYSICAL VIROLOGY IN TRIESTE
19.07.2017
Yesterday, our fellow Luca Tubiana presented his talk “Influence of Mutations on the Compactness of Viral ssRNA Genomes. A Detailed Computational Study of MS2 and BMV RNA2” at the Workshop on Physical Virology, organized by International Centre For Theoretical Physics of Unesco IAEA in Trieste (Italy).
You can watch Luca’s talk here.
NEW PUBLICATION
18.07.2017
The INDICAR-Fellow Mohamed Elgendy has co-authored the paper “PP2A Controls Genome Integrity by Integrating Nutrient-Sensing and Metabolic Pathways with the DNA Damage Response” published in the journal Molecular Cell. Congratulations!
Mec1ATR mediates the DNA damage response (DDR), integrating chromosomal signals and mechanical stimuli. We show that the PP2A phosphatases, ceramide-activated enzymes, couple cell metabolism with the DDR. Using genomic screens, metabolic analysis, and genetic and pharmacological studies, we found that PP2A attenuates the DDR and that three metabolic circuits influence the DDR by modulating PP2A activity. Irc21, a putative cytochrome b5 reductase that promotes the condensation reaction generating dihydroceramides (DHCs), and Ppm1, a PP2A methyltransferase, counteract the DDR by activating PP2A; conversely, the nutrient-sensing TORC1-Tap42 axis sustains DDR activation by inhibiting PP2A. Loss-of-function mutations in IRC21, PPM1, and PP2A and hyperactive tap42 alleles rescue mec1 mutants. Ceramides synergize with rapamycin, a TORC1 inhibitor, in counteracting the DDR. Hence, PP2A integrates nutrient-sensing and metabolic pathways to attenuate the Mec1ATR response. Our observations imply that metabolic changes affect genome integrity and may help with exploiting therapeutic options and repositioning known drugs.
TALKS AT SACRE COEUR HIGH SCHOOL
21.06.2017
On Monday 19th of June two INDICAR-Fellows, Cornelia Rumpf-Kienzl and Luca Tubiana, visited Sacre Coeur High School. They presented the 6A and 5E students their research in cancer and talked about everyday life of scientist as well. These talks developed the students’ interest in research careers. Thanks Sacre Coeur for having us, it was a great experience!



NEW PUBLICATION
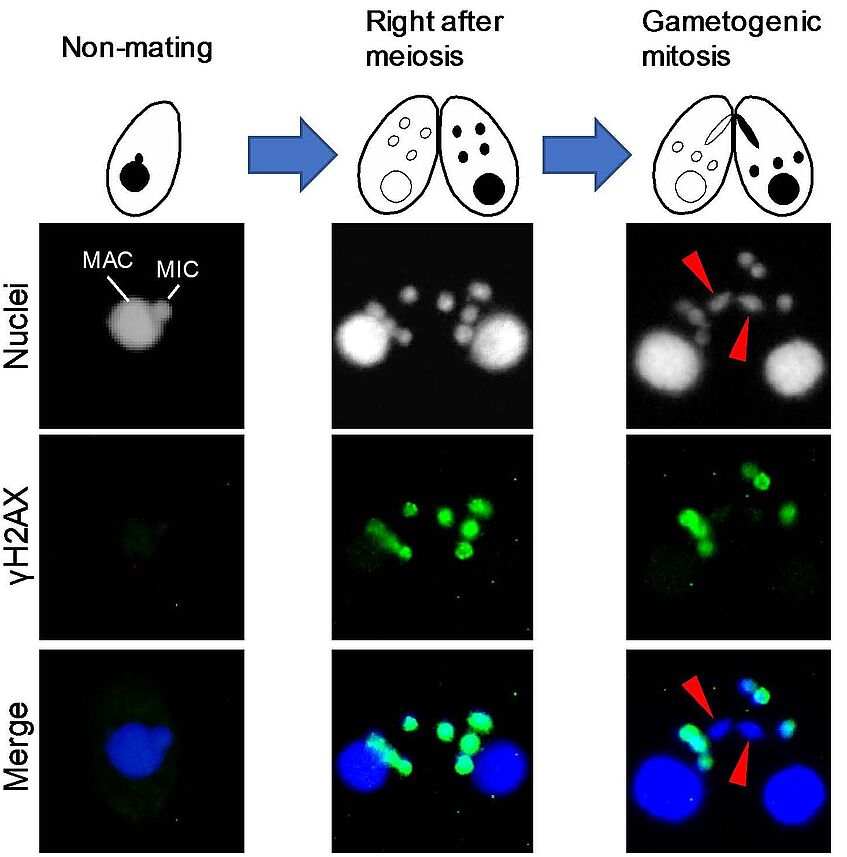
NEW PUBLICATION
19.06.2017
The INDICAR fellow Takahiko Akematsu just published the paper "Post-meiotic DNA double-strand breaks occur in Tetrahymena, and require Topoisomerase II and Spo11" in eLife. Congratulations!
https://elifesciences.org/articles/26176
Based on observations of markers for DNA lesions, such as phosphorylated histone H2AX (γH2AX) and open DNA ends, it has been suggested that post-meiotic DNA double-strand breaks (PM-DSBs) enable chromatin remodeling during animal spermiogenesis. However, the existence of PM-DSBs is unconfirmed, and the mechanism responsible for their formation is unclear. Here, we report the first direct observation of programmed PM-DSBs via the electrophoretic separation of DSB-generated DNA fragments in the ciliate Tetrahymena thermophila. These PM-DSBs are accompanied by switching from a heterochromatic to euchromatic chromatin structure in the haploid pronucleus. Both a topoisomerase II paralog with exclusive pronuclear expression and Spo11 are prerequisites for PM-DSB induction. Reduced PM-DSB induction blocks euchromatin formation, characterized by histone H3K56 acetylation, leading to a failure in gametic nuclei production. We propose that PM-DSBs are responsible for histone replacement during the reprogramming of generative to undifferentiated progeny nuclei.
NEW PUBLICATION
12.06.2017
The INDICAR-Fellow Nicola Silva contributed to Dr. Verena Jantsch work published in Genetics:
“Nuclear Envelope Retention of LINC Complexes Is Promoted by SUN-1 Oligomerization in the Caenorhabditis elegans Germ Line”.
Abstract: SUN (Sad1 and UNC-84) and KASH (Klarsicht, ANC-1, and Syne homology) proteins are constituents of the inner and outer nuclear membranes. They interact in the perinuclear space via C-terminal SUN-KASH domains to form the linker of nucleoskeleton and cytoskeleton (LINC) complex thereby bridging the nuclear envelope. LINC complexes mediate numerous biological processes by connecting chromatin with the cytoplasmic force-generating machinery. Here we show that the coiled-coil domains of SUN-1 are required for oligomerization and retention of the protein in the nuclear envelope, especially at later stages of female gametogenesis. Consistently, deletion of the coiled-coil domain makes SUN-1 sensitive to unilateral force exposure across the nuclear membrane. Premature loss of SUN-1 from the nuclear envelope leads to embryonic death due to loss of centrosome–nuclear envelope attachment. However, in contrast to previous notions we can show that the coiled-coil domain is dispensable for functional LINC complex formation, exemplified by successful chromosome sorting and synapsis in meiotic prophase I in its absence.
NEW PUBLICATION
06.06.2017
Our fellow Antje Koller published the paper “Testing the Implementation of a Pain Self-Management Support Intervention for Oncology Patients in Clinical Practice: A randomized Controlled Pilot Study”, in the journal Cancer Nursing. Congratulations Antje!
Abstract:
Background: In oncology, pain control is a persistent problem. Significant barriers to cancer pain management are patient related. Pain self-management support interventions have shown to reduce pain intensity and patient-related barriers. Comparative effectiveness research is a suitable approach to test whether effects are sustained in clinical practice.
Objective: In this pilot randomized controlled trial, the implementation of the ANtiPain intervention into clinical practice was tested to assess the effects on pain intensity, function-related outcomes, self-efficacy, and patient-related barriers to pain management to prepare a larger effectiveness trial.
Methods: Within 14 months, 39 adult oncology patients with pain scores of 3 or higher on a 10-point numeric rating scale were recruited in an academic comprehensive cancer center in Southern Germany. Patients in the control group (n=19) received standard care. Patients in the intervention group (n=20) received ANtiPain, a cancer pain self-management support intervention based on 3 key strategies: provision of information, skill building, and nurse coaching. An intervention session was performed in-hospital. After discharge, follow-up was provided via telephone calls. Data were collected at baseline and 1 and 6 weeks after discharge. Effect sizes were calculated for all outcomes.
Results: Large effects were found for activity hindrance (Cohen d=0.90), barriers (d=0.91), and self-efficacy (d=0.90). Small to moderate effects were found for average and worst pain (Cohen d=0.17-0.45).
Conclusions: Key findings of this study involved function-related outcomes and self-efficacy.
Implications for Practice: Because these outcomes are particularly meaningful for patients, the integration of ANtiPain to routine clinical practice may be substantial. A larger study will be based on these findings.
MC Fellowship
Our fellow Alessio Terenzi was granted a Marie Curie fellowship under the H2020-MSCA-IF-2016 call. After he has finished his INDICAR fellowship, he is going to join Prof. Luca Salassa (Inorganic Photochemistry Lab) at the Donostia International Physics Center / University of The Basque Country (San Sebastian, Spain). Congratulations!
NEW PUBLICATION
Alessio Terenzi published in the Journal Dalton Transactions: "Self-assembled Pt2L2 boxes strongly bind G-quadruplex DNA and influence gene expression in cancer cells" pubs.rsc.org/en/Content/ArticleLanding/2017/DT/C6DT03876J
Furthermore, Alessio Terenzi was awarded the front cover of the first issue of Dalton Transaction in 2017: pubs.rsc.org/en/content/articlepdf/2017/dt/c7dt90006f

NEW PUBLICATION
NEW PUBLICATION
Mohamed Elgendy (http://indicar.univie.ac.at/fellows/elgendy-mohamed/) just published an article in the Journal of Clinical Investigation (Impact Factor 13.2): "Dual modulation of MCL-1 and mTOR determines the response to sunitinib" https://www.jci.org/articles/view/84386
"In brief, the paper unravels mechanisms by which tumors adapt to and resist a drug called Sunitinib that's the first line therapy (best option) for renal cancer, making almost all the patients treated with this drug relapse within months. We also identified a clinical approach to make those tumors sensitive to this drug and potentially expand the lifespan of those patients."
This publication was also featured in one of the biggest newspapers in Italy: http://www.repubblica.it/salute/2016/11/28/news/tumori_resistenza_ai_farmaci_ieo-153021807/
INDICAR Workshop 2016
INDICAR - Workshop took place in Palermo, Italy, from Monday 12th Sept until Friday 16th Sept 2016.
The programme and timetable can be found on the webpage of the Workshop.

NEW PUBLICATION
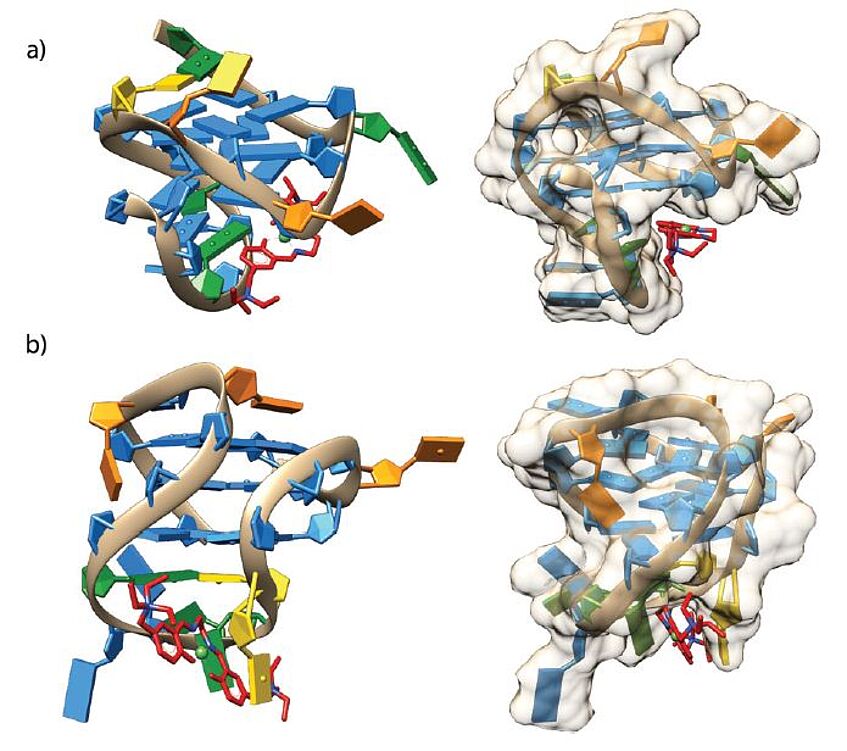
NEW PUBLICATION
Our INDICAR fellow Alessio Terenzi just published the paper "Another step toward DNA selective targeting: NiII and CuII complexes of a Schiff base ligand able to bind gene promoter G-quadruplexes" in Dalton Transactions.
Abstract: DNA G-rich sequences are able to form four-stranded structures organized in stacked guanine tetrads. These structures, called G-quadruplexes, were found to have an important role in the regulation of oncogenes expression and became, for such a reason, appealing targets for anticancer drugs. Aiming at finding selective G-quadruplex binders, we have designed, synthesized and characterized a new water soluble Salen-like Schiff base ligand and its NiII and CuII metal complexes. UV-Vis, circular dichroism and FRET measurements indicated that the nickel complex can stabilize oncogene promoter G-quadruplexes with high selectivity, presenting no interactions with duplex DNA at all. The same compound exhibited dose-dependent cytotoxic activity in MCF-7 breast cancer cells when combined with lipofectamine as lipophilic carrier.
http://pubs.rsc.org/en/content/articlelanding/2016/dt/c6dt00648e
Interview

Interview
Two of our INDICAR fellows (Laura Cimatti and Anne Conibear) gave an interesting interview to the UNI:VIEW Magazin of the University of Vienna. Click here to read the interview (in German).
